Calamari Clots Part 2: The Elemental Chemical Puzzle
Elemental analysis reveals a matrix unlike any conventional thrombus
In Part 1 of this series, we discussed the first in a research trilogy by Bruce Rapley and Matt Shelton, sponsored by New Zealand Doctors Speak Out. That initial report focused on the gross morphology and microscopic histology of anomalous intravascular casts (AICs), also referred to as “calamari clots.” These are the white, rubbery structures that embalmers have reported regularly since the onset of the campaign to inject the world’s population with COVID-19 spike protein–producing genetic products in the name of “vaccination.”
Building on that foundation, the researchers demonstrated that, “based on morphology and histology, the anomalous intravascular casts exhibited a consistent set of features that distinguish them from both classical antemortem thrombi and postmortem coagula.” In other words, they are fundamentally different from both normal blood clots that form during life or after death. “Their elasticity, lumen-conforming geometry, cohesive fiber networks, sparse cellular composition, and partial but genuine Lines of Zahn collectively indicate a formation process that does not align with established thrombotic pathways.” While morphology alone cannot determine causation, these reproducible features strongly suggest that AICs may represent a previously unclassified intravascular entity.
Now we will explore part 2 of the Rapley and Shelton trilogy, which is titled: Elemental Characterization of Anomalous Intravascular Casts Reveals an Abnormal Biochemical Matrix.
Please consider supporting our work with a paid subscription, or a one-time donation.
What is “Elemental Analysis”?
Before we look at the research involving the elemental chemistry of complex structures such as the AICs and the clotting protein fibrinogen, lets use some very simple molecules to demonstrate the concepts of elemental analysis.
Please recall your basic high school chemistry. Molecular theory holds that all things are composed of molecules, and all molecules are composed of atoms. Consider the period table.
The periodic table presents all known and theoretical atoms according to their mass and the corresponding size and nature of their outer electron shells. Each atom’s size and electron configuration determines the ways it may combine and react with other atoms. The nature of atoms determines the molecules which they can form.
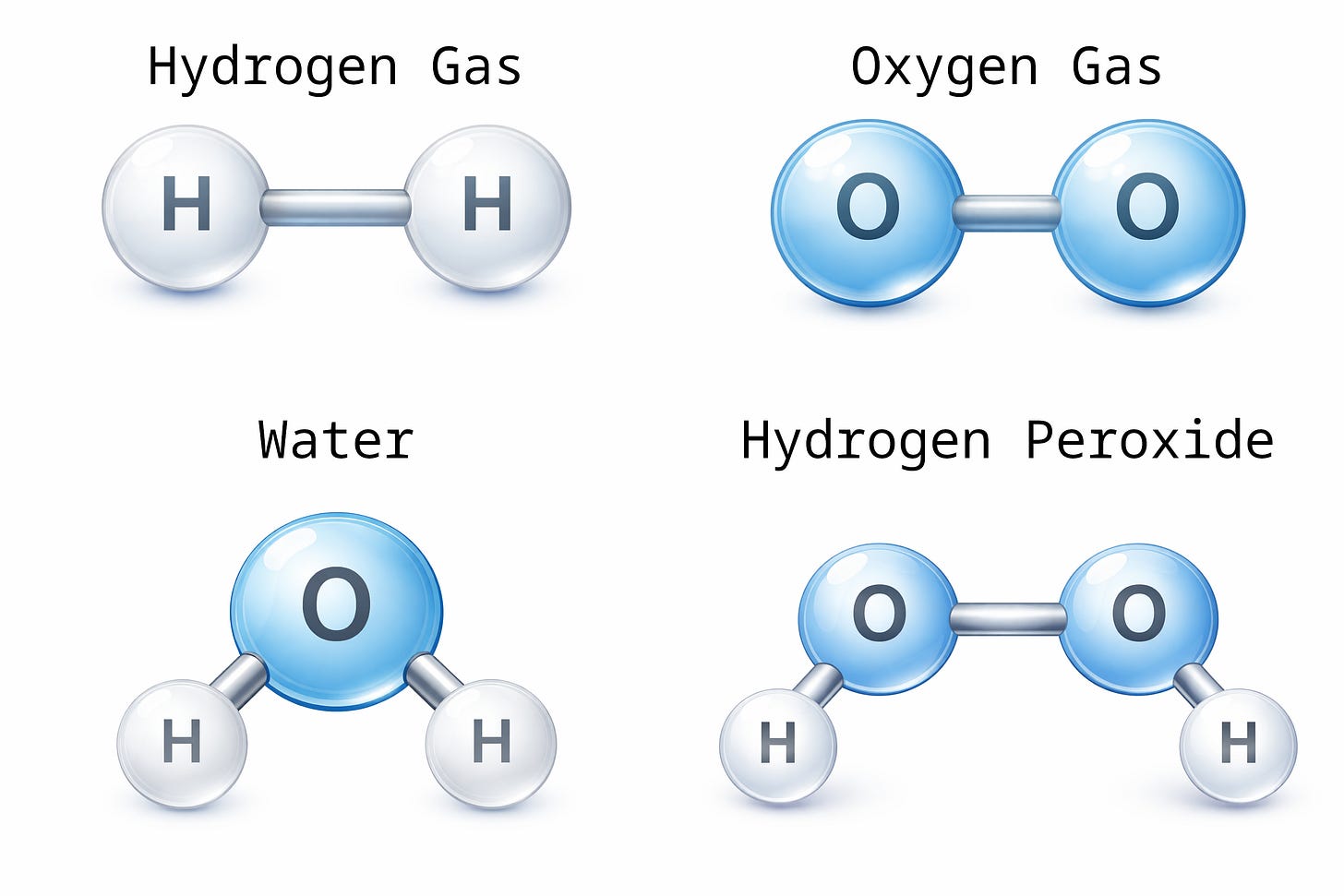
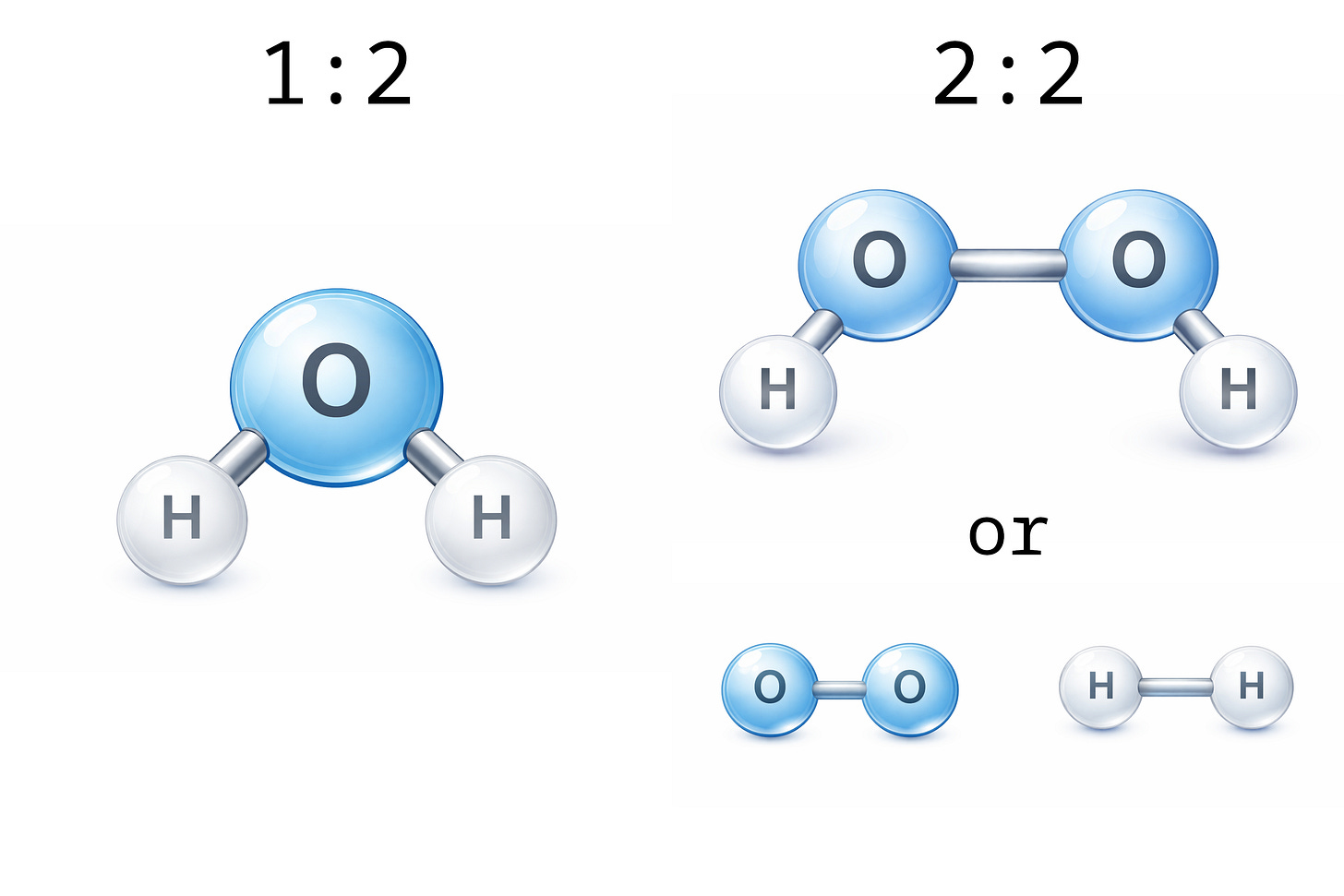
For instance, consider hydrogen atoms and oxygen atoms. Two hydrogen atoms combined into one molecule renders one molecule of hydrogen gas. Two oxygen atoms combined into one molecule renders one molecule of oxygen gas. One oxygen atom and two hydrogen atoms combined into one molecule render H2O, or “water.” Two oxygen and two hydrogen atoms combined into one molecule render hydrogen peroxide.
If I tell you that a substance is composed of oxygen and hydrogen atoms, it could be water, it could be hydrogen peroxide, or it could be a mixture of oxygen gas and hydrogen gas; but it could not be methane. Methane molecules are each composed of four hydrogen atoms combined with one carbon atom. Similarly, a substance whose atomic elements are limited to hydrogen and oxygen could not be table salt, because salt crystals are composed of sodium and chloride atoms in a one-to-one ratio.
To demonstrate the utility of measuring the ratio of the atoms in a substance, consider that a ratio of 2 hydrogen atoms per oxygen atom is consistent with water, while a ratio of 1 hydrogen atom per 1 oxygen atom is consistent with hydrogen peroxide, but it is also consistent with an equal mixture of oxygen (O₂) and hydrogen (H₂) gas; however, it is not consistent with water (H₂O).
Determining what atoms a substance is made of, and the ratio of those atoms, does not tell you the definitive chemistry of the substance. It does not tell you exactly what molecules the substance is made of, but still a lot can be deduced from the elemental composition. For instance, if two substances are the same, they will have the same atomic elements in the same ratios.
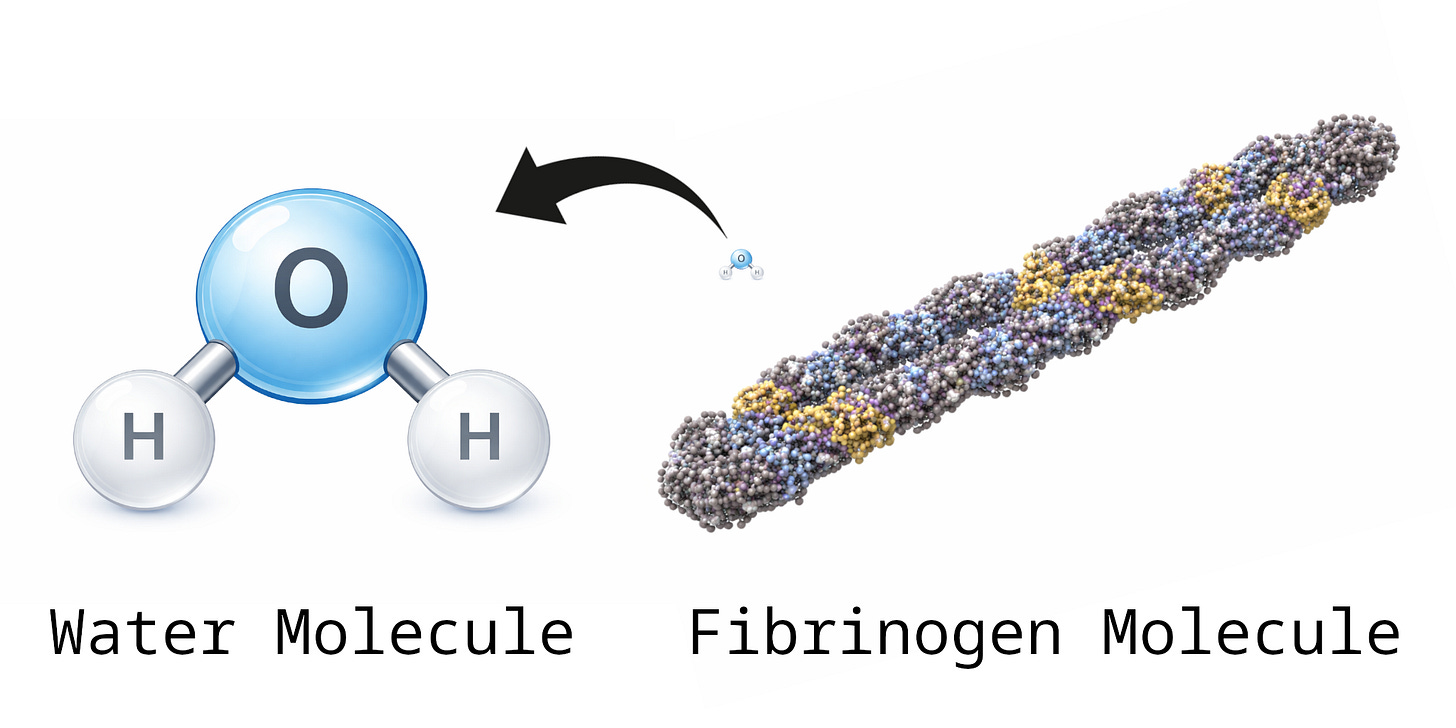
The principles for studying elemental chemistry that are demonstrated in these simple examples of water and methane also apply to the elemental analysis of more complex structures like anomalous intravascular casts (“calamari clots”) or complex proteins like the clotting protein fibrinogen. To demonstrate the complex math involved in this sort of analysis, consider the size and structure of fibrinogen. Fibrinogen is the fundamental protein from which normal blood clots are made. Soon you will see how the predicted elemental composition of fibrinogen is involved in the analysis of anomalous intravascular casts in this study.
With regards to the size and complexity of a fibrinogen molecule, consider this:
A hydrogen atom has an atomic weight of 1 Dalton; an oxygen atom is 16 Daltons, a water molecule H₂O is 18 Daltons, a carbon atom is 12 Daltons, and a methane molecule CH₄ is 16 Daltons. Fibrinogen is so much larger than these simple molecules that there does not exist a simple chemical formula for it. One fibrinogen molecule includes more than 15,000 carbon atoms, 24,000 hydrogen atoms, 4,000 nitrogen atoms, 5,000 oxygen atoms, and 100 sulfur atoms. There are various slightly different forms of fibrinogen, their molecular weights varying from 340,000 to 420,000 Da.
A very crude attempt at a molecular formula for fibrinogen would look something like C₁₅₀₀₀H₂₄₀₀₀N₄₀₀₀O₅₀₀₀S₁₀₀ or higher.
Though the analysis is more complex than our examples with simple molecules like water and methane, a variety of insights and conclusions can be drawn from the accurate atomic elemental composition analysis of AICs which is found in this study.
The Technique Used: ICP-MS
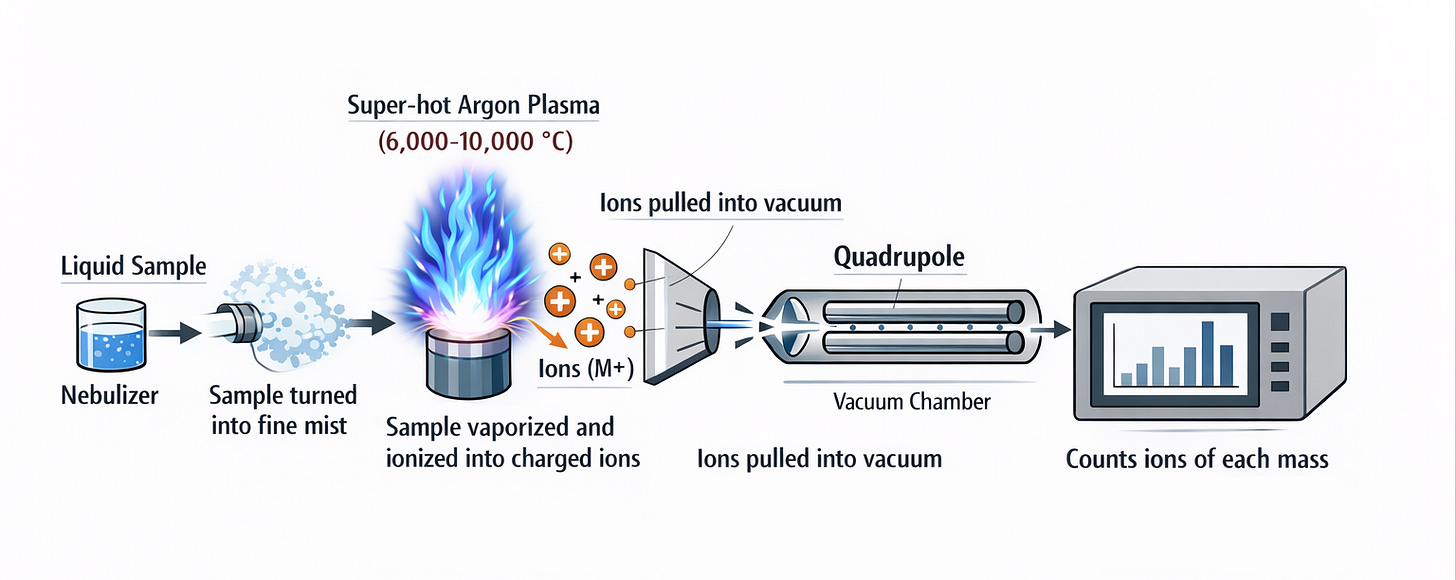
The researchers used Inductively Coupled Plasma Mass Spectrometry (ICP-MS).
Here is the essence of how Inductively Coupled Plasma Mass Spectrometry works:
First, the sample is turned into a fine mist and sprayed into a super-hot flame called plasma. This plasma is made by heating argon gas with radio waves to temperatures hotter than the surface of the sun, around 6,000–10,000 °C. The intense heat breaks the sample apart into individual atoms and strips off electrons, turning the atoms into positively charged ions, or the nuclei of the atoms
These ions are then pulled into a vacuum chamber, where a mass spectrometer acts like a super-accurate scale. It separates the ions based on their mass (how heavy they are) and charge. Lighter ions bend more in electric or magnetic fields, while heavier ones bend less. A detector counts the ions of each type.
By knowing which masses appear and how many, scientists can identify which elements are present and their exact amounts.
The Samples
In the current study, the researchers analyzed the same intravascular cast specimens examined in the first paper of the series.
To maintain scientific rigor:
Samples were coded and blinded
Multiple laboratories performed analyses
Labs were not told what the samples were
This prevents subconscious bias from influencing measurements.
After the elemental results were finalized, they were compared with:
Theoretical values for fibrin-based clots
Measure values for whole blood
Previously measure values for “Adam’s Blood” (one 2022 un-vaccinated whole blood sample)
Previously measured values for “Adam’s Clot” (one 2022 AIC from a dead Covid-19 “vaccinated” individual)
“Adam’s clot” and “Adam’s whole blood” refer to specific ICP-MS analyzed samples in a 2022 report by Mike Adams.
In Mike Adams’ 2022 tests, the sample size was very small: one calamari clot for Adam’s Clot, and one blood sample for “Adam’s blood.” There are variations in which elements were and were not specifically tested for in Adams’ 2022 tests and the current study, so you will note some blanks in some of the comparison tables.
By comparing the elemental composition results for the AICs in the study to those of Adam’s blood, Adam’s clot, and theoretical values for fibrinogen based on its known molecular structures, the researchers sought to determine whether the unusual casts matched known blood clot elemental chemistry and to gain other insights into the AICs’ chemical structure.
A little more preamble:
Before we review the researchers’ results and analysis, please allow me to explain a few other terms encountered in their work.
Fibrinogen is a large protein that is abundant in our blood. It is a stable, water-soluble protein until it is activated by cleaving small sections from its ends. This renders the fibrinogen active, and many molecules of activated fibrinogen join together in a fibrous structure, which is the foundation and main substance, along with platelets and red blood cells, of normal blood clots.
“Fibrinogen Stoichiometric Value” refers to the elemental composition predicted for fibrinogen based on this protein’s established average chemical structure (“average” because there are variations of fibrinogen due to glycosylation and various isoforms that differ significantly).
The Study’s Findings:
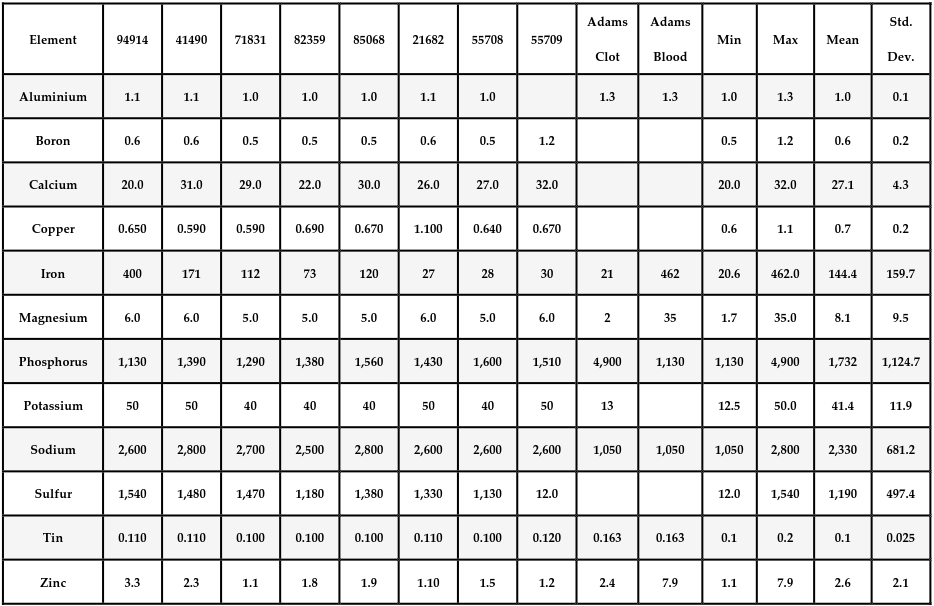
The samples were specifically tested for their levels of the elements aluminum, boron, calcium, copper, iron, magnesium, phosphorus, potassium, sodium, sulfur, tin, and zinc. Carbon, hydrogen, nitrogen, and oxygen were not directly tested.
The following table summarizes the current study’s elemental composition results for eight different AICs that were tested. They are presented along with the 2022 Mike Adams results for one AIC (Adam’s clot) and one non-C19 “vaccinated” healthy individual’s whole blood (Adam’s Blood). (table below)
The table above summarizes average elemental concentrations for eight anomalous casts studied (as well as reference values for “Adam’s Blood” and “Adam’s Clot”). Concentrations are in parts per million.
The summary table above was created using the results of a series of ICP-MS analyses for various atomic elements. Below are those series of analyses, which were compiled to create the master table above.
Note the consistency among all eight of the AICs studied. The patterns are quite different from whole blood and are not fully consistent with Mike Adams’ 2022 analysis of a single AIC. Note that the missing bars for Adam’s Blood and Adam’s Clot in these graphs occur because Mike Adams’ analysis in 2022 did not include those atomic elements in the case of boron and calcium, or because the levels where much lower in the case of copper.
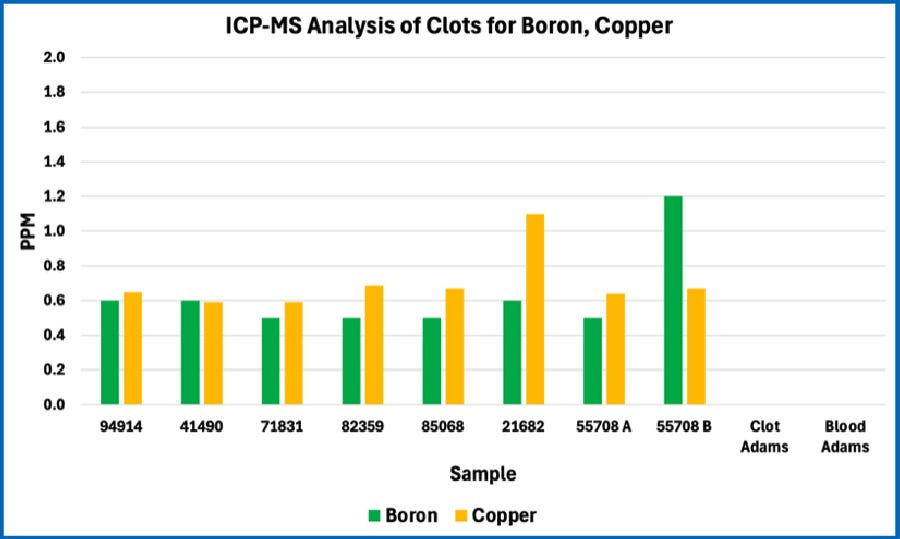
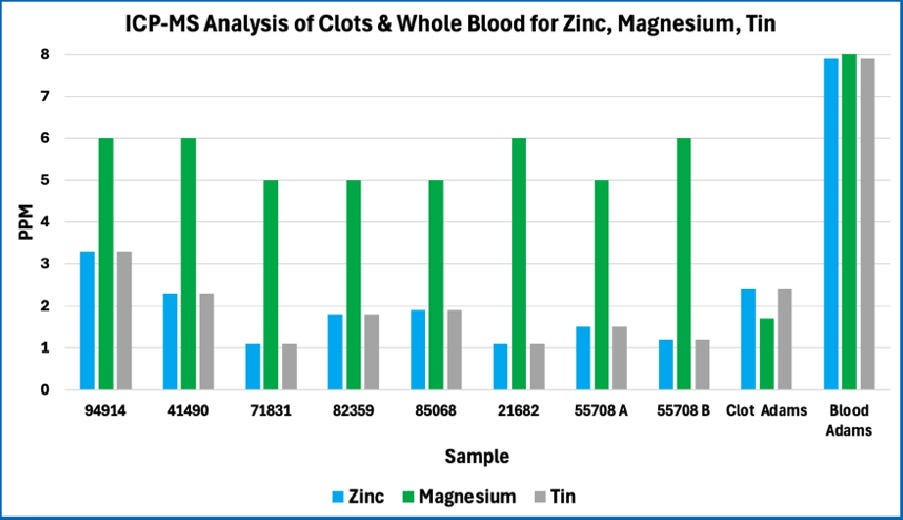
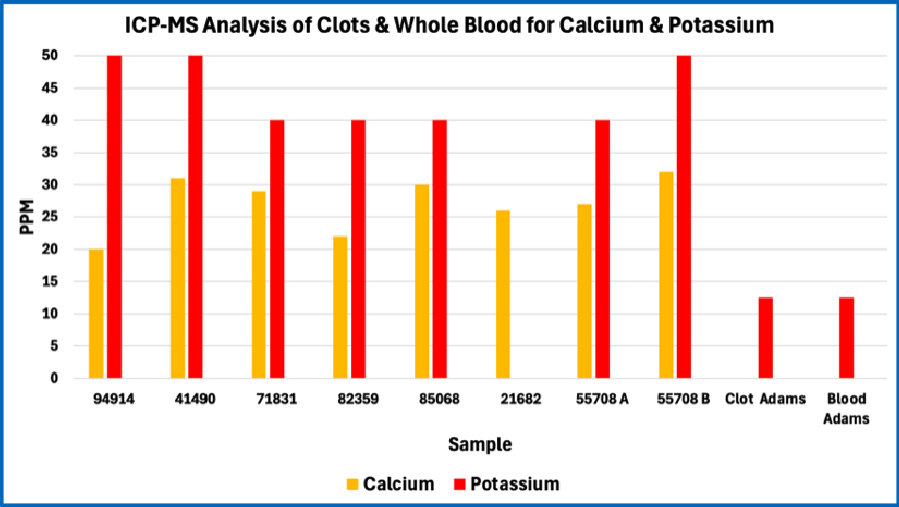
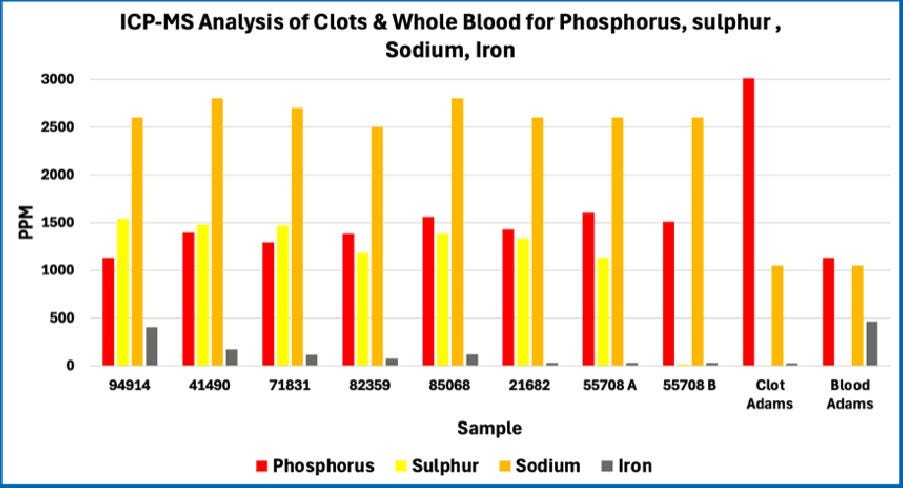
Consistency among the AICs studied
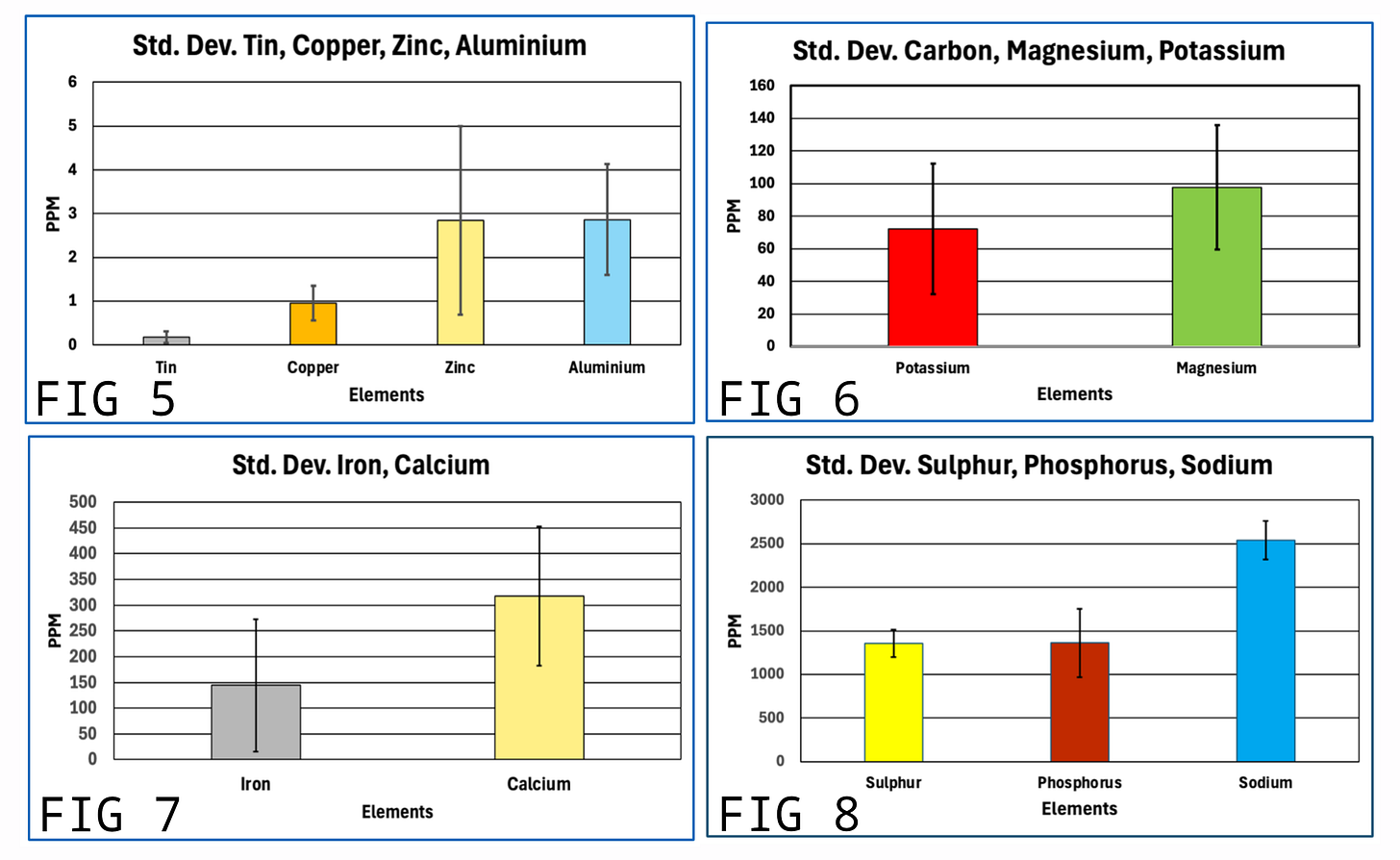
The figures above reveal strong consistency in the composition of the eight AICs studied in terms of the atomic elements that were tested. This indicates that the AICs are not random structures. Rather, this is consistent with AICs having a recurring structure and composition. This is further demonstrated by the relatively small standard deviation in results among the eight AICs; this is shown in the four figures below.
These charts confirm that although individual values fluctuate, the overall elemental signature remains stable.
Let’s look at one more chart from the study before we move into the analysis, conclusions, theories, and questions that can be drawn from the work. This table compares these elemental composition values: 1. measured composition of whole blood; 2. calculated composition for fibrinogen, based on its known chemistry; and 3. the mean values for AICs from this study.
The table also includes ratios for these elements between the AICs tested and whole blood, and between the AICs tested and the predicted values for fibrinogen. Blanks in the fibrinogen column are elements that are not expected in fibrinogen and could be replaced with “approximately zero.” Blanks in the experimental clots column are elements that were not directly tested in this experiment (carbon, hydrogen, nitrogen, and oxygen). Blanks in the ratios columns occur where there is no calculable ratio because either the concentration of that element was not measured in the AICs studied, or that element has a predicted concentration of ~0 in fibrinogen.
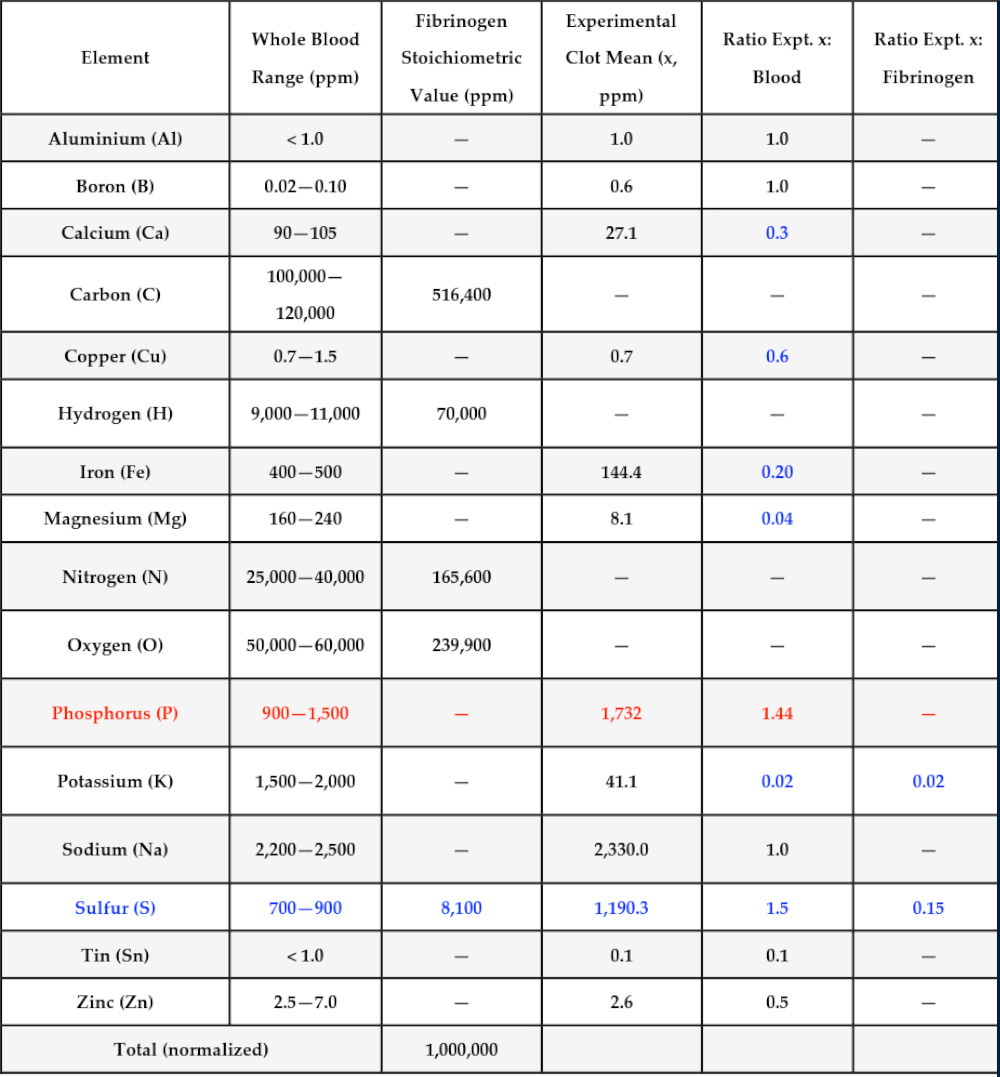
Note that, compared to whole blood, the AIC (“clots”) studied are significantly lower in calcium, copper, iron, magnesium, potassium, and sulfur, but are significantly higher in phosphorus.
Compared to predictions for natural physiologic fibrinogen-based clots, the AICs are very high in phosphorus and very low in sulfur.
Analysis, Conclusions, Theories and Questions:
First there are three key chemical differences identified that distinguish these casts from normal fibrinogen blood clots:
1. Sulfur depletion
Sulfur is a key element in proteins, particularly in the amino acids cysteine and methionine. Because fibrin is itself a protein, normal blood clots contain relatively predictable amounts of sulfur that reflect the amino acid composition of fibrinogen, the precursor molecule that forms fibrin fibers during coagulation. In a matrix composed primarily of fibrin, sulfur should account for roughly 0.8% of the total mass.
However, the casts analyzed in this study contained far less sulfur than expected, measuring approximately one-eighth of that concentration. This substantial depletion indicates that, despite their fibrin-like appearance under the microscope, the material cannot be composed predominantly of protein and must include a significant proportion of non-protein constituents.
2. Elevated phosphorus
At the same time, the casts exhibited higher-than-expected levels of phosphorus. Phosphorus is commonly found in several important biological molecules, including phospholipids that make up cell membranes, nucleic acids such as DNA and RNA, inorganic polyphosphate chains, and phosphorylated proteins that carry phosphate groups as part of cellular signaling and regulation.
In contrast, fibrin itself contains relatively little phosphorus. The expected fibrinogen stoichiometric value of phosphorus is approximately zero. The elevated phosphorus levels observed in these casts therefore suggest that the matrix includes additional phosphate-bearing components beyond fibrin, pointing toward the presence of non-protein constituents within the material.
3. Nitrogen
The researchers discuss a third, and in my opinion weaker, finding related to nitrogen. Please recall that in this experiment the researchers did not test the samples directly for nitrogen, carbon, oxygen, or hydrogen. However, the researchers used analytic methods that inferred that the significant non-protein, non-fibrinogen portion of the casts, as indicated by the measured sulfur depletion, was relatively deficient in nitrogen relative to oxygen and carbon. This is consistent with there being significant non-protein elements in the AICs. This lends further support to the sulfur findings, which indicate that much of the AICs are not protein.
What Does This Mean?
Taken together, these chemical findings suggest that, though under the microscope the casts include fibrous elements, they are hybrid materials. Protein, which may indeed be fibrinogen, can account for a significant part of the material, but it does not dominate the structure. The casts contain a mixture of components, including substances that are richer in phosphorus (as measured) and oxygen (as calculated).
Consistency Across Samples
One of the strongest aspects of the findings is reproducibility. Despite differences in sample size, dehydration levels, and handling conditions, the same chemical patterns appeared repeatedly. The relative ordering of major elements remained consistent across laboratories. This consistency suggests the AICs’ compositional phenomenon is not random contamination or a rare anomaly.
What the Elemental Data Rules Out
The elemental chemical findings allow several possible explanations for the existence of AICs to be excluded:
1. Excess Fibrin
If these casts were simply very large clots made mostly of fibrin, sulfur levels would be high. They are not. Therefore, fibrin overproduction alone cannot explain the structures.
2. Packed Blood Cells
If the casts were mainly composed of compressed blood cells, the material would show high levels of nitrogen, sulfur, and iron. Instead, nitrogen appears low, sulfur is depleted, and iron levels are modest. This rules out the idea that the casts are just compacted cellular debris.
3. Postmortem artifacts
After death, blood chemistry changes and clots can form. However, postmortem processes do not selectively remove sulfur while increasing phosphorus. Because the same pattern appears repeatedly across samples and laboratories, the researchers conclude the AICs material is not simply a postmortem artifact.
Possible Sources of the Phosphorus
Fibrin alone cannot explain the chemistry. This raises the question: “What contributes the abundant phosphorus found in the AICs?” The researchers outline several possibilities:
Membrane fragments
When red blood cells or platelets break apart, their membranes release phospholipids. These molecules contain phosphorus in their head groups. Large amounts of membrane fragments could increase phosphorus levels while contributing little sulfur. However, the relatively low levels of iron in the AICs indicate that there is a relative absence of the iron-rich red-blood-cell protein hemoglobin.
Platelet polyphosphate
Activated platelets release chains of inorganic phosphate known as polyphosphate. These molecules can bind to fibrin and alter clot structure. Polyphosphate could therefore increase phosphorus content without dramatically affecting sulfur levels.
Phosphorylated proteins
Proteins can carry phosphate groups through chemical modifications called phosphorylation. Although these modifications add phosphorus, they typically occur in small numbers and are unlikely to account for the entire phosphorus excess.
Viral or inflammatory interactions
Some studies have shown that certain viral proteins can interact with clotting proteins and modify clot structure. For example, laboratory research has reported interactions between SARS-CoV-2 spike protein and fibrin. However, the elemental data alone cannot confirm whether such interactions play a role in the casts. Further molecular analysis is required.
What Elemental Analysis Cannot Tell Us
Although elemental chemistry reveals important clues, it has limits. Elemental measurements cannot identify specific proteins, molecular structures, or biochemical pathways.
Knowing that phosphorus is present does not reveal whether it comes from disrupted cell membrane phospholipids, platelet polyphosphate, phosphorylated proteins, or other processes such as spike protein interaction with fibrin.
To answer those questions, researchers must examine the molecules themselves.
Why the Next Step Is Proteomics
Because the elemental analysis shows that the casts are not simple fibrin clots, the logical next step is to determine exactly which proteins are present.
Proteomic analysis can identify:
Individual proteins
Their relative abundance
Chemical modifications
This will reveal whether the protein scaffold of the casts resembles normal clotting biology or something fundamentally different. That molecular investigation forms the subject of the third and final paper in this research trilogy by Bruce Rapley and Matt Shelton.
The Takeaway:
The elemental study provides strong evidence that anomalous intravascular casts represent a distinct biological material.
Key findings include:
Dramatic sulfur depletion
Significant phosphorus enrichment
Reduced protein dominance
Consistent elemental patterns across samples
These chemical constraints show that the casts cannot be explained by ordinary clotting processes.
Combined with the structural observations reported in the first paper, the evidence suggests that these formations represent a previously uncharacterized intravascular matrix.
Exactly which molecules create this matrix remains unknown.
However, the chemical clues uncovered in this study narrow the possibilities and set the stage for the final investigation.
In Part 3, researchers turn to proteomics to identify the specific proteins and molecular components responsible for these unusual intravascular structures.
We are grateful to the researchers Bruce Rapley and Math Shelton, and their sponsors at New Zealand Doctors Speak Out, for advancing our understanding of the white rubbery anomalous intravascular casts. These advances will help us improve patient treatment and save lives.
Footnote: AICs are not the only clotting pathology that follow the COVID-19 genetic injections. The science discussed in my Jan 2024 video and article titled, “New Science: How SARS CoV-2 and the Genetic Jabs Cause Blood Clots; New research shows how the engineered SARS-CoV-2 virus, as well as the genetic jabs, sabotage blood cell properties that help fight natural coronaviruses to cause deadly blood clots instead,” remains relevant in explaining red blood cell clumping and many clot disorders that have arisen in COVID-19 “vaccination” victims.








I had a hard time following THE SCIENCE, but this is what I do know. My cousin was pretty much unable to walk. This was in the fall of 2023. He had major blood clots in his legs. They got removed. I year later, he was diagnosed with a rare leukemia and died in mid December of 2024. He was in his mid 70's.
"previously unclassified intravascular entity..." However, from your work Mark, I could tell what was going on when my father got his first fakeVax, and had a stroke the next day. My mother could never walk again after her fakeVax the same day, telling me her legs always hurt, which I figured were caused by some kind of "clots". Later, their MD's knew the vax killed them, but was fired for telling the truth, allowing the murdering USgov to illegally alter their death certificates to "natural causes". They tucked them away as a "win" for their lying, insane, subhuman existence, so their deaths were not even counted as vaxDeaths to date.